First Experimental Decoupling of Interfacial Proton Transport in Energy Device Materials --A New Method for Quantifying Ion Transport at Electrode Interfaces--
  |
First Experimental Decoupling of Interfacial Proton
Transport in Energy Device Materials
-- A New Method for Quantifying Ion Transport at Electrode Interfaces--
Key Points
- First successful separation of proton transport at different interfaces for energy device materials
- Previously overlapped transport components were experimentally decoupled
- Enables accurate and quantitative evaluation of interface-specific proton conductivity
| A research team led by Yusuke Abe (Former master student), Dr. Kentaro Aoki, Dr. Athchaya Suwansoontorn, and Professor Yuki Nagao at the Materials Chemistry Frontier Research Area, Japan Advanced Institute of Science and Technology (JAIST), in collaboration with the University of Calgary (Professor Kunal Karan) and Tokyo University of Science (Associate Professor Isao Shitanda), has developed a new measurement method that enables separation and quantification of proton transport at different interfaces in polymer electrolyte thin films. The team focused on interfaces between polymer electrolytes and electrode materials such as platinum and carbon. Conventional measurements have been unable to distinguish whether proton conductivity differs at each interface, because contributions from multiple interfaces are superimposed into a single component. By combining structural control of interdigitated electrodes with extended low-frequency impedance measurements, the researchers successfully separated and quantified these overlapping components. This study experimentally demonstrates, for the first time, that proton transport at different interfaces can be independently evaluated. This advancement significantly improves the understanding of ion transport at electrode interfaces and is expected to contribute to the performance improvement of a wide range of electrochemical devices. |
Background
In electrochemical devices such as fuel cells and water electrolyzers, polymer electrolytes (ionomers) coat electrode surfaces and transport protons across their interfaces. However, real systems involve multiple interfaces, including platinum and carbon surfaces. In conventional measurements, all contributions are combined into a single component, making it impossible to distinguish how much each interface contributes to proton transport.
This limitation has hindered the establishment of rational design guidelines for electrode-electrolyte interfaces.
Research Findings
The research team developed a new methodology by combining:
1. Structural control of interdigitated electrodes (IDEs, Figure 1)
2. Extended low-frequency electrochemical impedance spectroscopy (EIS)
This approach enabled the separation of previously overlapping transport components.
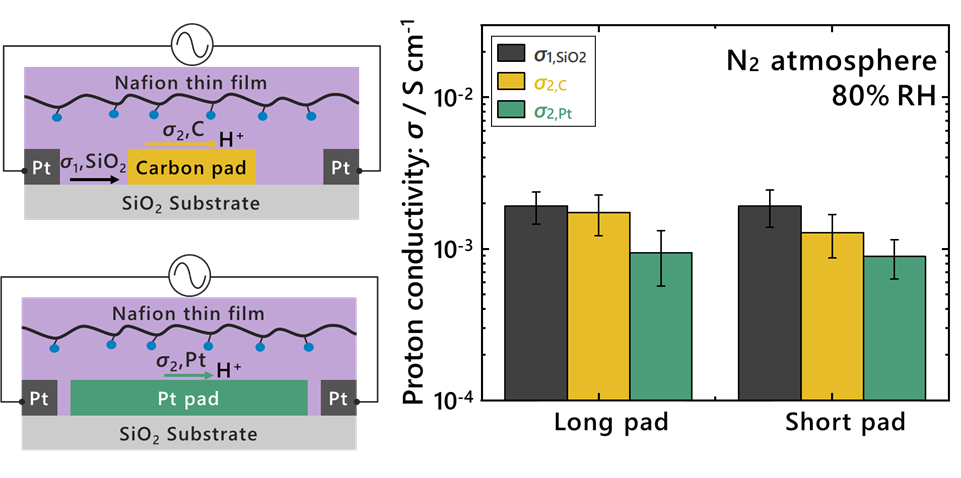
| Figure 1. Conceptual illustration and summary of the results in this study. The left panels illustrate proton transport along electrode interfaces within a Nafion thin film. Distinct transport pathways exist at the SiO₂ substrate interface (σ₁) and at the carbon or Pt electrode interfaces (σ₂). The right panel shows that the extracted conductivities (σ₁ and σ₂) remain nearly identical even when the electrode pad length (Long / Short) is varied. This indicates that the measured conductivities are intrinsic to each interface rather than dependent on electrode geometry, demonstrating that interfacial proton conductivity can be quantified with improved accuracy. |
The study revealed that:
1. The high-frequency component corresponds to proton transport at the SiO₂ interface
2. The low-frequency component corresponds to proton transport at Pt and carbon interfaces
Furthermore, the extracted conductivities were independent of electrode geometry, confirming that the measured values represent intrinsic interface-specific properties. This demonstrates that proton conductivity, previously measured as a single averaged value, can now be separated and quantified for each interface with improved accuracy.
Future Prospects
This study establishes a methodology for evaluating proton transport at individual electrode-electrolyte interfaces. The ability to identify which interface limits ion transport will enable more rational design of electrode structures and material selection. The method is applicable not only to fuel cells but also to a wide range of electrochemical devices, including water electrolysis systems, secondary batteries, and sensors.
Future work will explore the applicability of this approach to more complex electrode systems and further clarify the relationship between interfacial structure and ion transport.
Funding
This research was supported by JST CREST (Grant No. JPMJCR21B3), JSPS KAKENHI grant number JP21H00020 (Hydrogenomics) and Murata Science and Education Foundation.
Glossary
A hydrogen ion (H⁺) that carries positive charge and plays a key role in electrochemical energy systems.
A polymer material that conducts ions. In this study, Nafion was used as a representative proton-conducting ionomer.
A widely used as a benchmark polymer electrolyte known for its high proton conductivity.
The boundary where two different materials meet, such as between polymer electrolyte and electrode surfaces.
A measure of how easily protons move through a material or along an interface.
A comb-like electrode structure used to measure in-plane ion transport.
A technique that analyzes material responses to AC signals to evaluate ion transport and resistance over a range of frequencies.
Publication Details
| Title: | Decoupling Interfacial Proton Conductivity in Ionomer Thin Films on Pt and Carbon Electrodes |
| Authors: | Yusuke Abe, Kentaro Aoki, Athchaya Suwansoontorn, Kunal Karan, Isao Shitanda, Yuki Nagao* |
| Affiliation: | Japan Advanced Institute of Science and Technology (JAIST), University of Calgary, Tokyo University of Science |
| Journal: | ACS Applied Materials & Interfaces |
| Publication Date: | May 1, 2026 |
| DOI: | 10.1021/acsami.6c04425 |
May 11, 2026
